THE ACS MAH SYSTEM OF THE PHARMACEUTICAL INDUSTRY
Counterfeit medicines are now appearing all over the world – including in Europe. Therefore, in 2011 the European Union decided to minimize the risk of counterfeit medicines entering the legal supply chain. Since February 2019, only prescription-only medicines marked with the new safety features may be marketed. The legally required verification of the medicines takes place via a special protection system.
THE PROTECTION SYSTEM FOR THE AUTHENTICITY CHECK
| MAH System | System of the pharmaceutical industry |
| NAMS | National Alert Management System |
| HP | Hospitals |
| PH | Pharmacies |
ACS MAH System
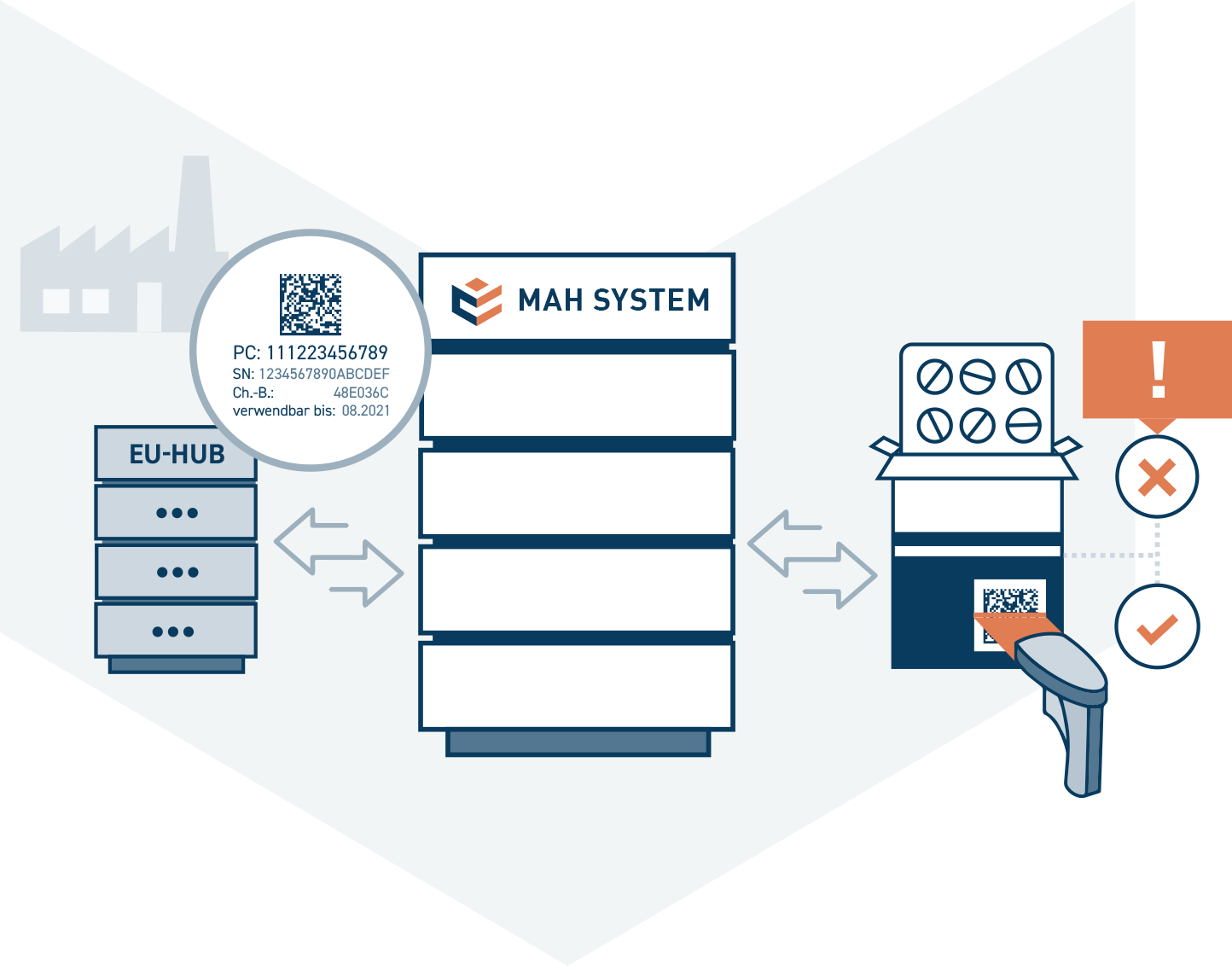
The ACS MAH system is the central database of the National Medicines Verification System in Germany, in which all pack data of drugs subject to verification are stored. The pack data consists of the product code, the batch number, the expiration date and the unique serial number. The manufacturer loads the pack data centrally into the ACS-PU system via the EU Hub.
This makes it possible to compare the data printed on a pack with the pack data stored in the system. This comparison is usually initiated by a wholesaler or pharmacy via the pharmacy system.
The system stores every query and status change of a drug pack. If it has the status “active” in the system, it can be dispensed. When a pack is dispensed, the system checks whether this pack is still “active”. If the check is successful, the status of the pack is changed to “inactive”. Data comparison prevents multiple packs with the same pack data from ending up in the hands of patients. If a discrepancy occurs during a query, an error message is automatically generated.
The ACS MAH System is a high-performance system that processes several million queries a day with a response time of around 30 milliseconds. Through the connection to the EU Hub, the system can also receive and process requests for pack data from other European countries. In addition, it is possible to use the ACS-PU system to forward requests for packs whose data is located in another national system via the EU hub.
Reporting System
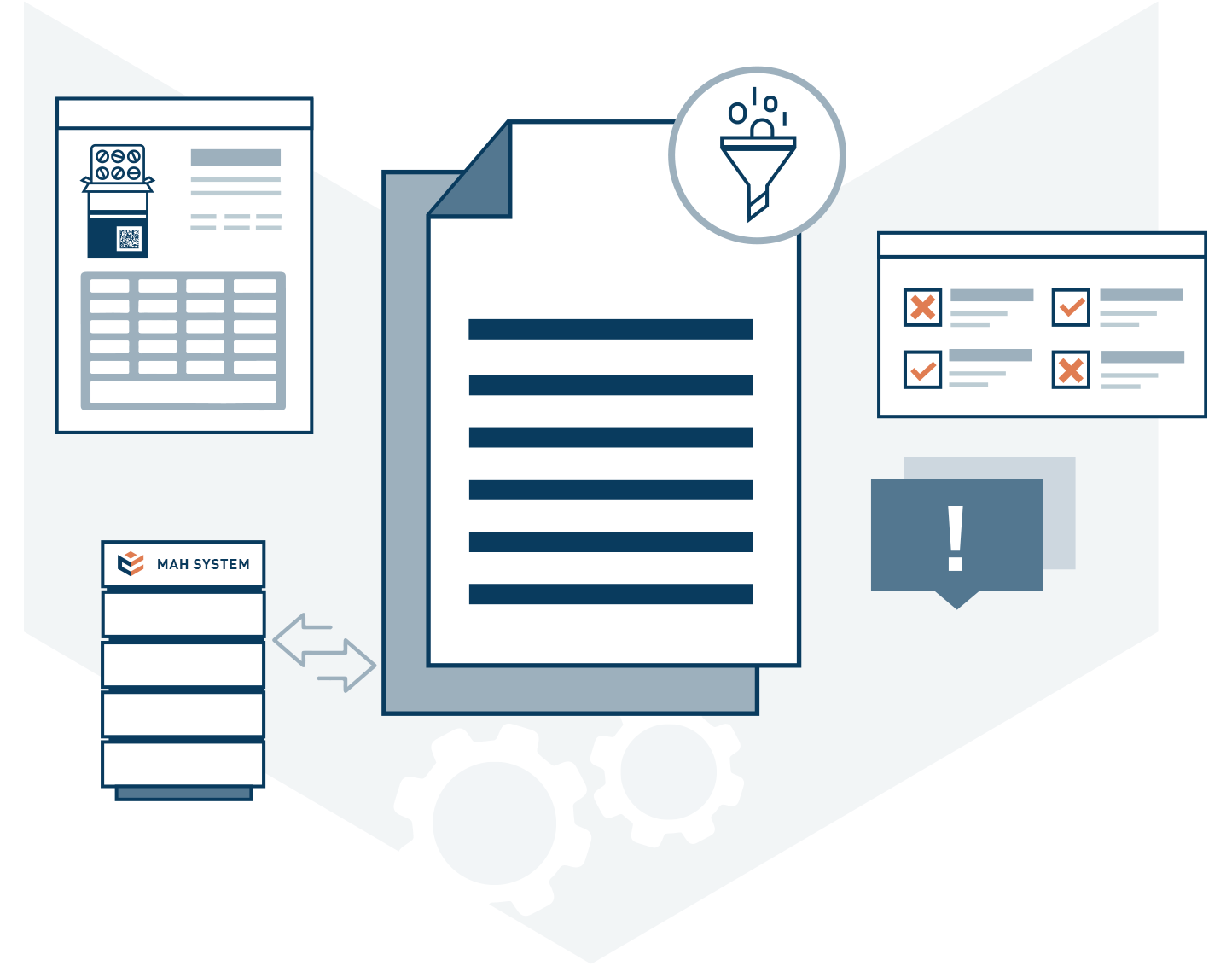
The reporting system offers various reports to our customers and to the authorities. For example, our customers can see which products they have uploaded and which batches with which number of packs for this product are available in the ACS-PU system. Additionally, the history of individual packs where an error message has occurred can be tracked. Each customer can create individual filters and views to get the specific information they need.
The reporting system loads the data daily from the ACS-PU system and prepares it for the various reports, so there is hardly any waiting time when using it.
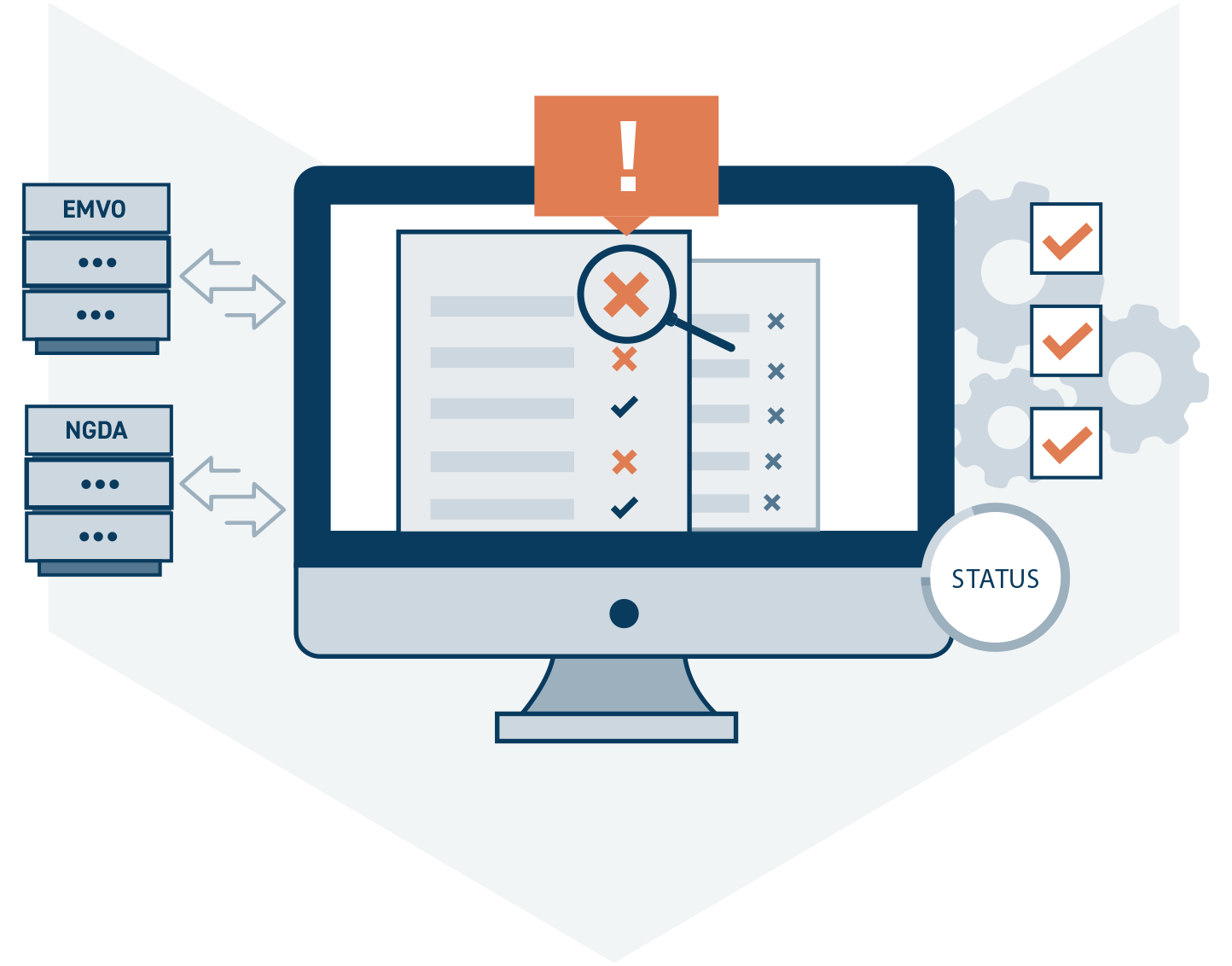
Alert-Management-System
The National Alert Management System (NAMS) collects and manages all error messages. Each customer thus receives the error messages for his own products. In addition, an initial system-based analysis is carried out to determine the error cause. Each error message has a status that describes its relevance. If an error message is escalated, it represents a potential case of suspected counterfeiting from the manufacturer’s point of view.
Error messages can also be classified as technical errors by automatic mechanisms in the NAMS and thus closed. In addition, each manufacturer can individually evaluate the relevance of error messages that have occurred for his own products.
The NAMS is connected to the AMS Hub of EMVO as well as to the Pharmacy system of NGDA. This makes it possible to obtain the findings of other parties involved in an error message and to share one’s own findings. In interaction with the data from the reporting system, the mandatory clarification of all error reports can be realized easily and quickly.